
WB optimization allowed the achievement of accurate results, and the easy DBS sample collection, transportation and storage will support the use of this immunodiagnostic technique for conducting HIV/acquired immunodeficiency syndrome (Aids) epidemiological surveys. To validate the WB testing, the reciprocal relationship between antibody reactivity of DBS samples and of the respective plasma samples was investigated. Results:Īll of the DBS samples showed the band pattern compatible with the criterion for HIV positivity established by the Ministry of Health. In order to assess the reliability of these results, the assay performance was previously certified by employing the sample panel (reference panel) of Instituto Adolfo Lutz (IAL), which consisted of whole blood and plasma paired samples. 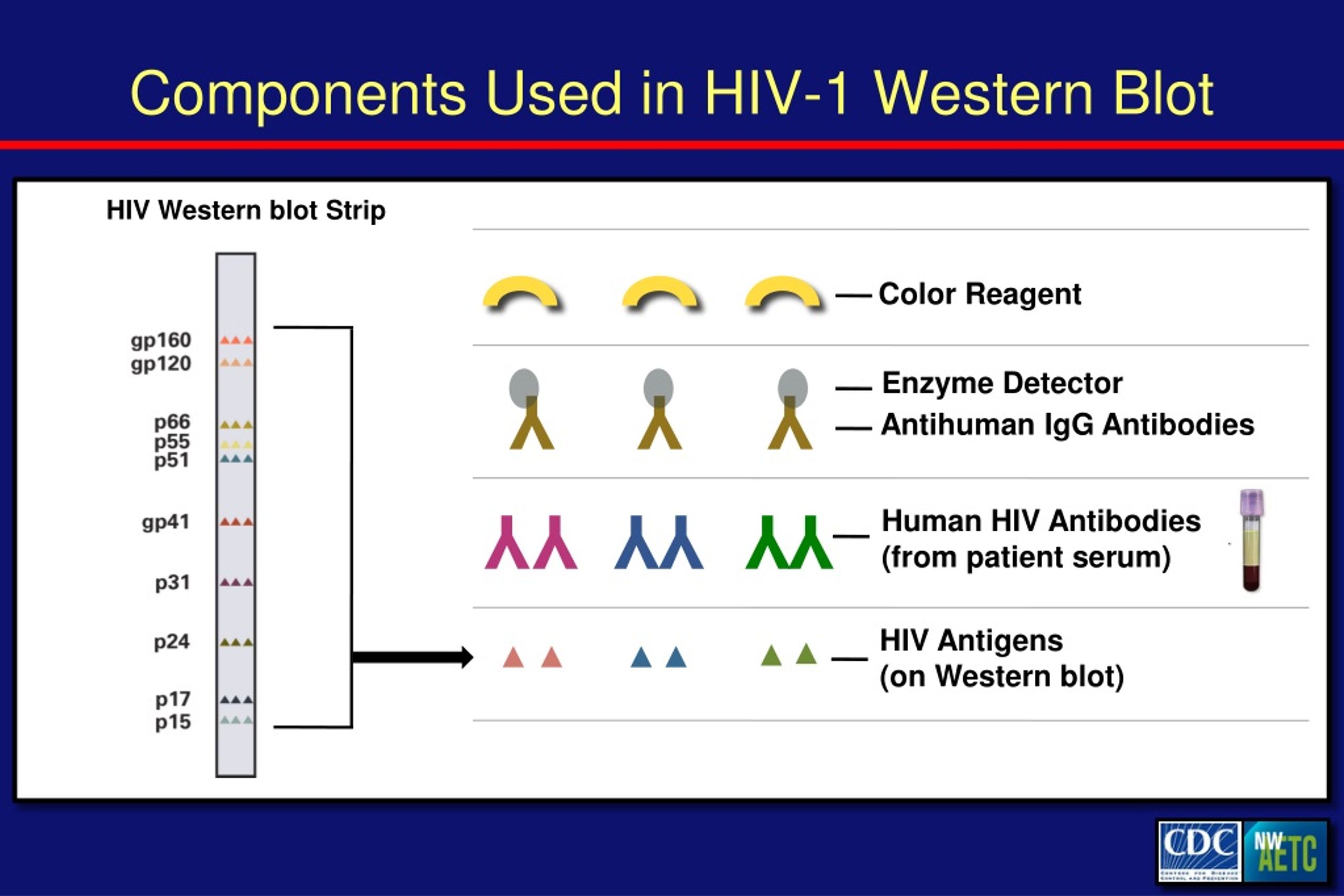
These samples derived from the SampaCentro Study, and they were HIV-antibody-positive in a screening test. One hundred eighteen blood samples collected in filter paper were analyzed by a modified WB. This study aimed at evaluating the analytical performance of WB assay for analyzing DSB samples. For HIV diagnosis, the conventional strategy firstly uses an enzyme immunoassay as screening test, and the positive sample is confirmed on a complementary assay, as western blotting (WB). Because of these characteristics, the use of DBS is recommended for studies in which the goal is to increase the population access to diagnostic testing, including HIV infection screening. For conducting field studies on human immunodeficiency virus (HIV) serodiagnosis, the use of samples collected on filter paper is profitable owing to the simplicity in handling and delivering them.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
